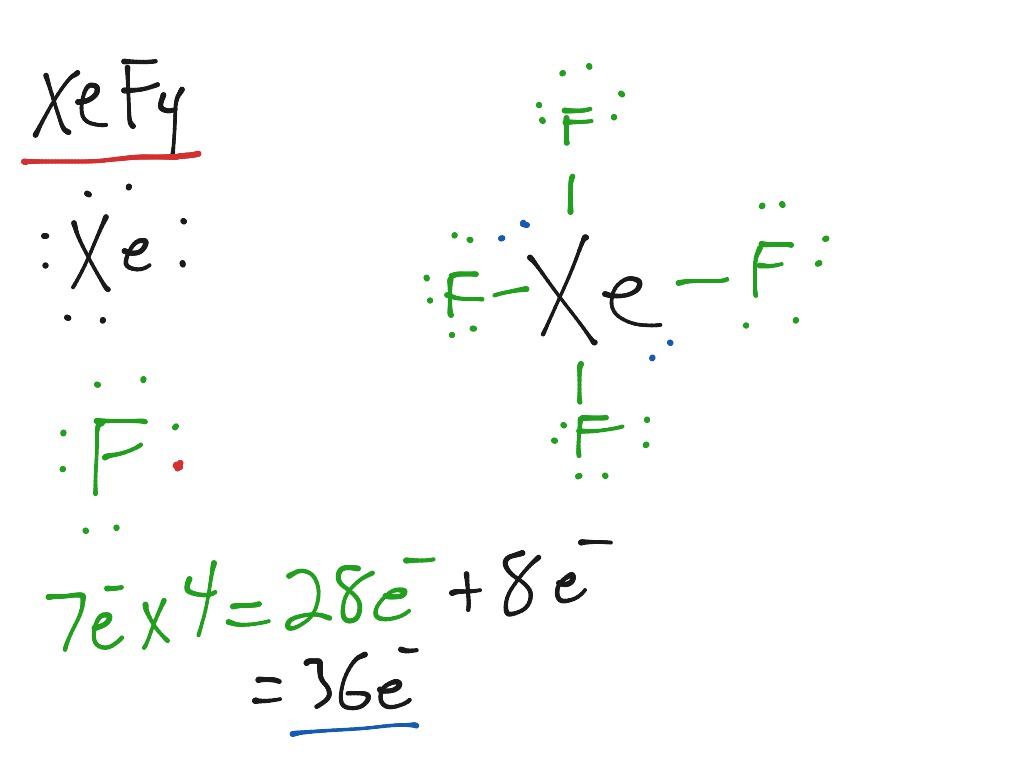
XeF4 dot structure Science ShowMe
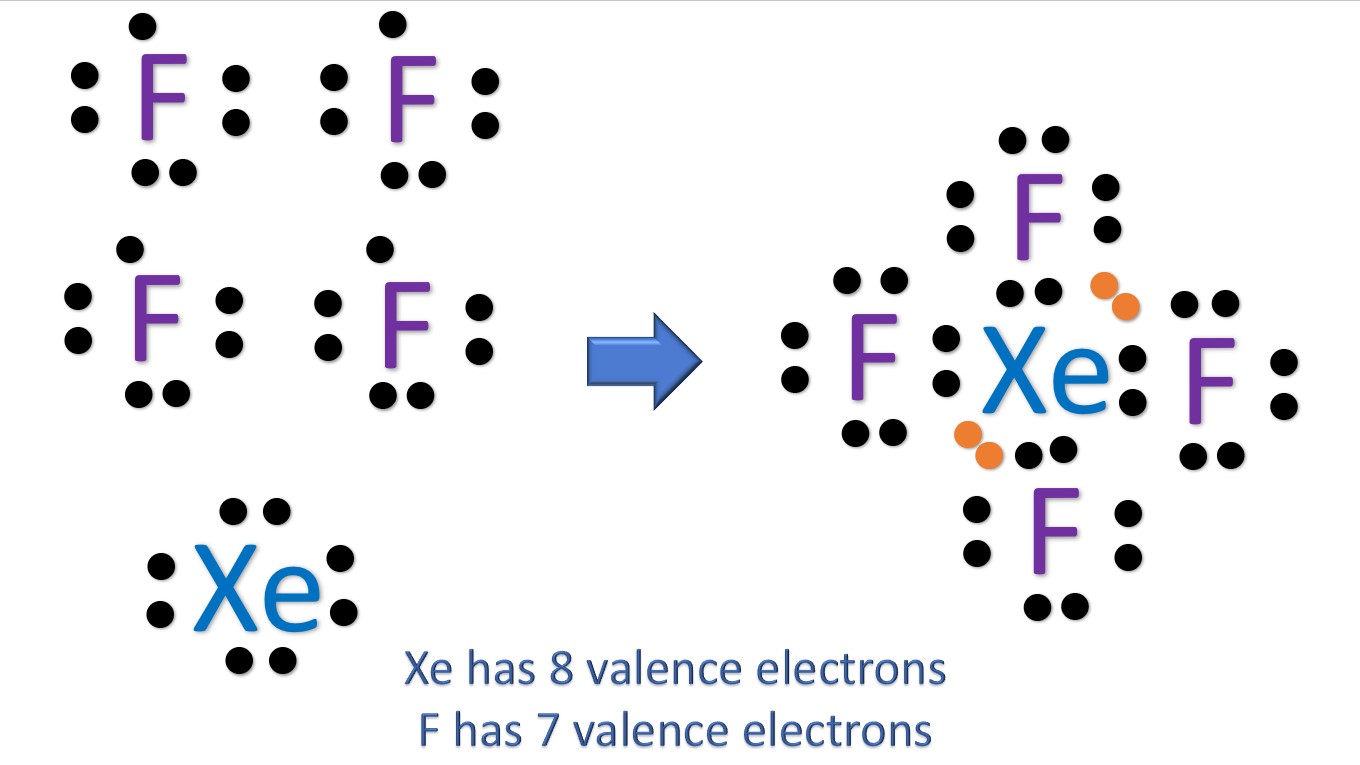
In the XeF 4 Lewis structure, there are four single bonds around the xenon atom, with four fluorine atoms attached to it. Each fluorine atom has three lone pairs, and the xenon atom has two lone pairs. XeF4 Lewis Structure - How to Draw the Lewis Structure for XeF4 Watch on Contents Steps #1 Draw a rough skeleton structure
Solved
XeF4 lewis structure has Xenon atom (Xe) at the center which is surrounded by four Fluorine atoms (F). There are 4 single bonds between the Xenon atom (Xe) and each Fluorine atom (F). There are 2 lone pairs on the Xenon atom (Xe) and 3 lone pairs on all the four Fluorine atoms (F).

So far, we’ve used eight of the XeF4 Lewis structure’s total 8
XeF4 is the chemical formula of the compound Xenon Tetrafluoride. This chemical compound is formed when xenon reacts with fluorine. Its chemical equation could simply be written as : Xe + 2F2 ——> XeF4 In this process, elemental fluorine supposedly oxidizes xenon, under some specific conditions of temperature and pressure.

Is XeF4 Polar or Nonpolar? Techiescientist
XeF4 Lewis Structure, Molecular and Electron Geometry based on the VSEPR theory, the steric number, Hybridization and expected bond angles.

Solved Molecule XeF4 has D4h symmetry. Draw its Lewis
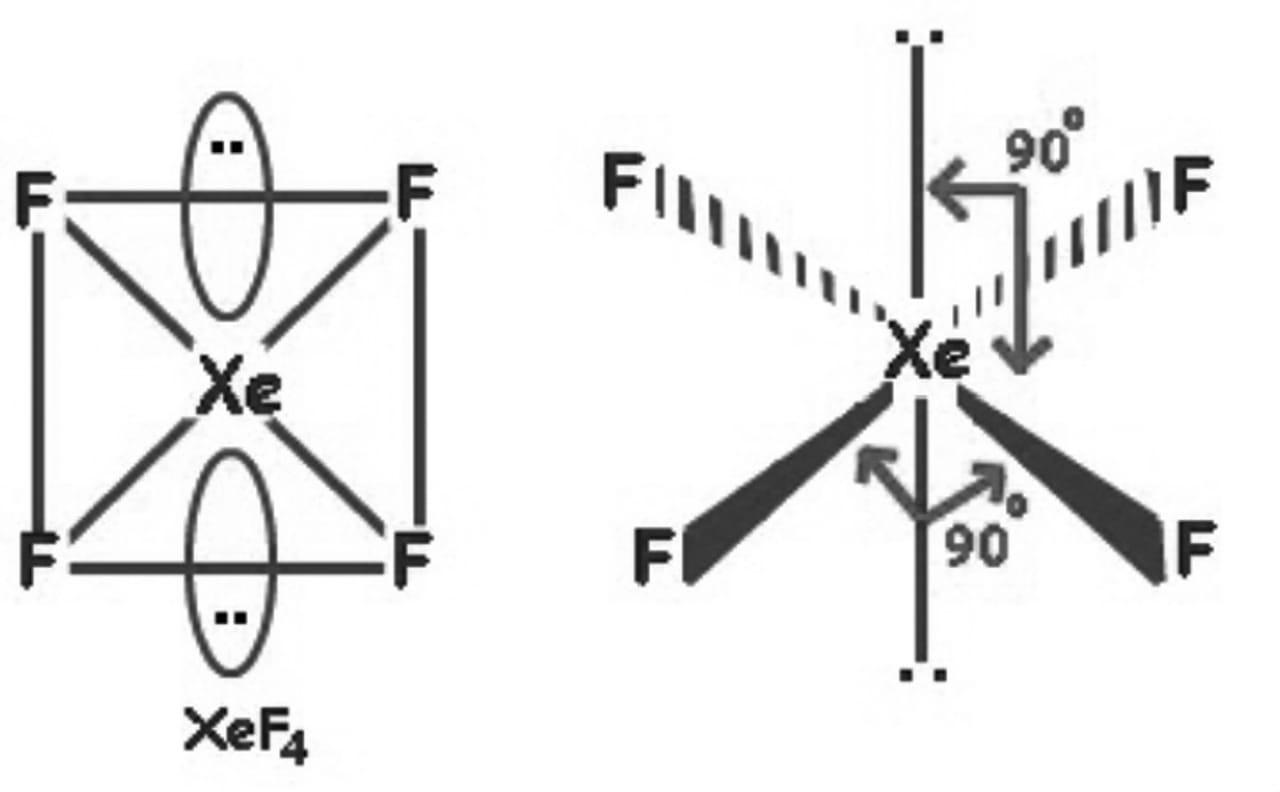
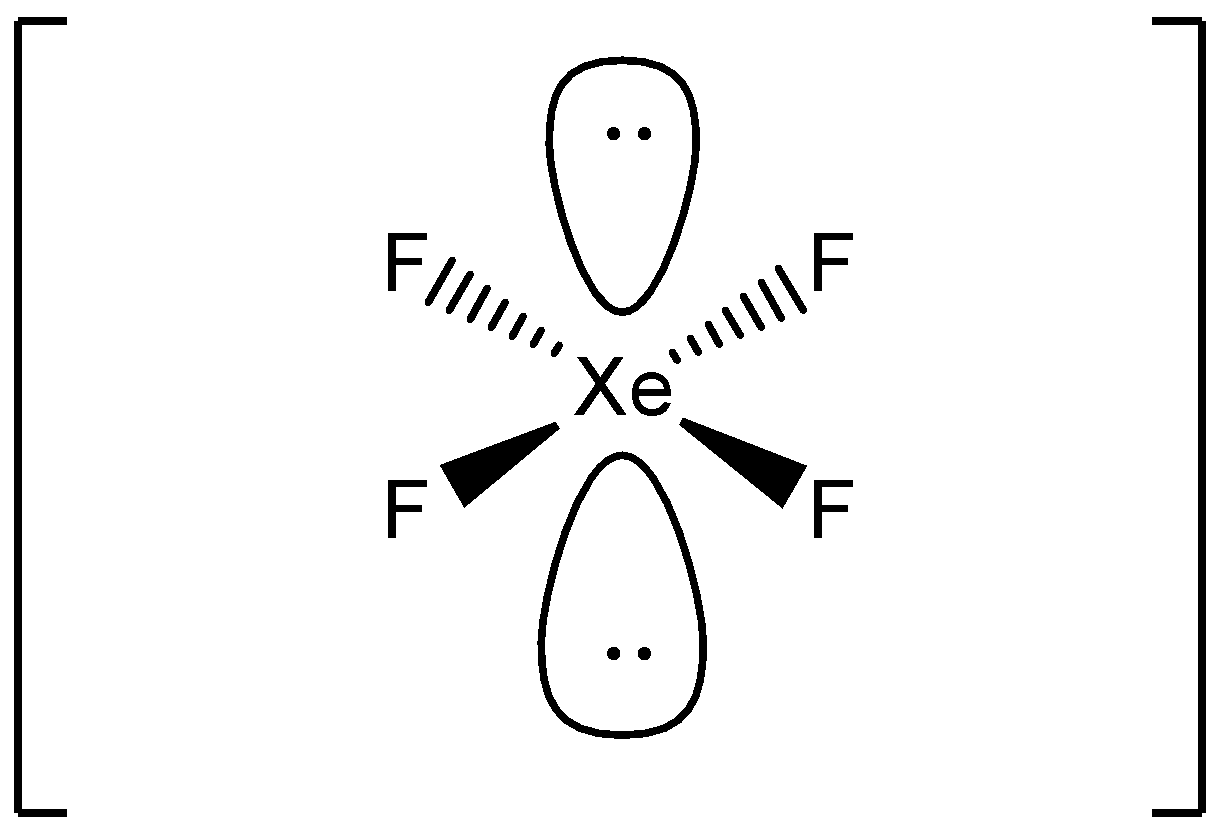
Xenon tetrafluoride (XeF4) is a square planar, non-polar molecule. The Xenon atom has 4 bonding pairs of electrons and 2 lone (non-bonding) pairs of electro.
Molecular Geometry of XeF4 [with video and free study guide]
Its structure was determined by both NMR spectroscopy and X-ray crystallography in 1963. [6] [7] The structure is square planar, as has been confirmed by neutron diffraction studies. [8] According to VSEPR theory, in addition to four fluoride ligands, the xenon center has two lone pairs of electrons. These lone pairs are mutually trans. Synthesis

Solved The Lewis structure for XeF4 is Xě Which of
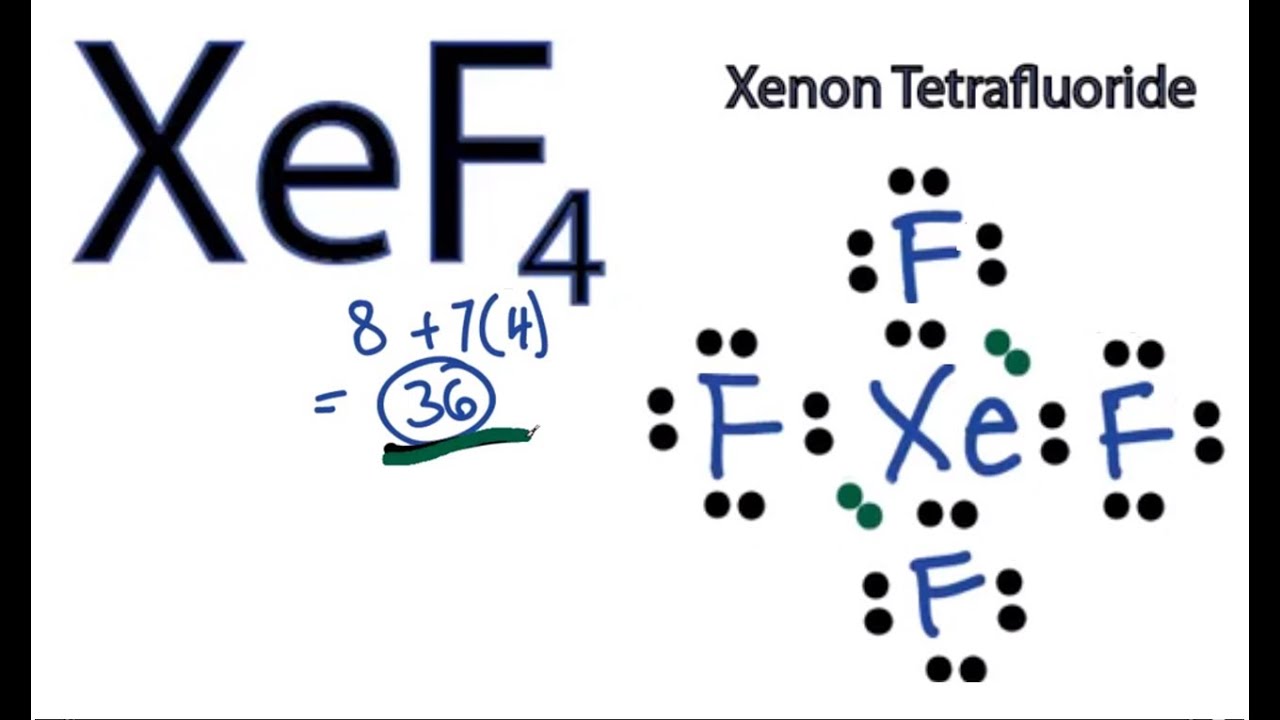
In the Lewis structure of XeF4 structure there are a total of 36 valence electrons. XeF4 is also called Xeon Tetrafluoride. Note that XeF4 can have an Expanded Octet and have more than.
XeF4 Lewis Structure How to Draw the Lewis Structure for XeF4 YouTube
In XeF 4 (Xenon tetrafluoride) lewis structure, there are four sigma bonds and two lone pairs around xenon atom. Each fluorine atom has three lone pairs. In this tutorial, we will learn how to draw lewis structure of XeF 4 step by step. Lewis structure of XeF 4

XeF4 Molecular Geometry, Bond Angles & Electron Geometry YouTube
Drawing the Lewis structure of XeF4 involves following a few steps. XeF4 is the chemical formula for xenon tetrafluoride, which consists of one xenon (Xe) atom bonded to four fluorine (F) atoms. Step-by-Step Guide to Drawing the Lewis Structure of XeF4 1. Count the total number of valence electrons
Molecular Geometry Of Xef4 Youtube
Lewis structure of XeF4 contains four single bonds between the Xenon (Xe) atom and each Fluorine (F) atom. The Xenon atom (Xe) is at the center and it is surrounded by 4 Fluorine atoms (F). The Xenon atom has 2 lone pairs and all the Fluorine atoms have 3 lone pairs. Let's draw and understand this lewis dot structure step by step.
[Solved] Complete the chart. Molecular Polarity. XeF4 +Lewis Structure
For the XeF4 Lewis structure we first count the valence electrons for the XeF4 molecule using the periodic table. Once we know how many valence electrons there are in XeF4 we can distribute them around the central atom and attempt to fill the outer shells of each atom. The Lewis structure for XeF4 has a total of 36 valence electrons.
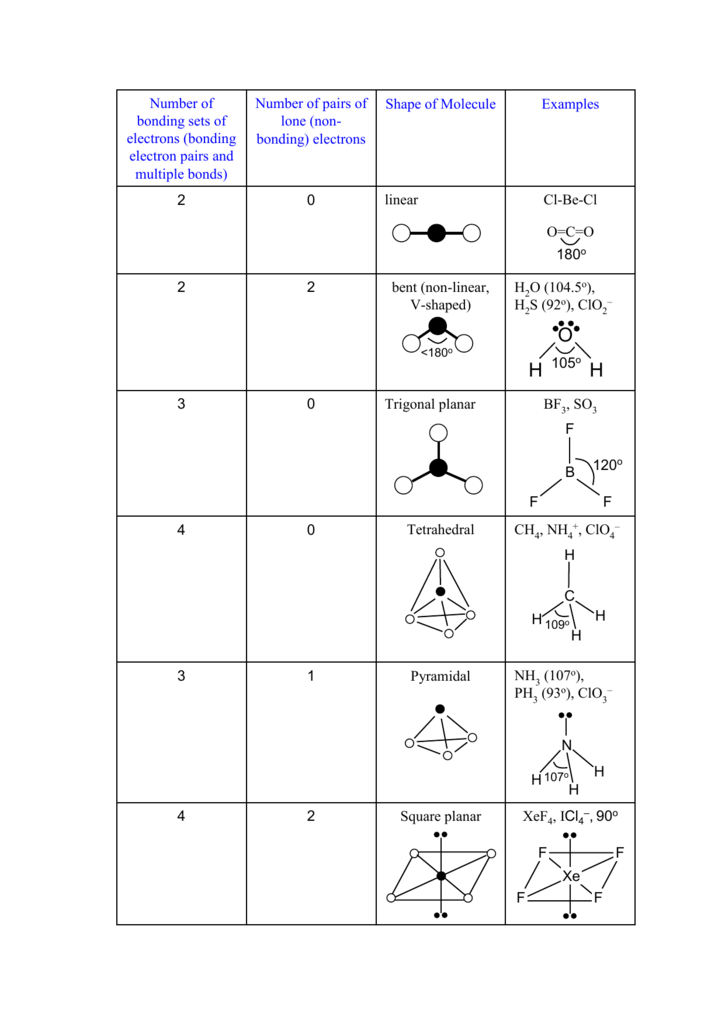
XeF4, ICl4 , 90o Square planar 2 4 NH (107o), PH (93o), ClO
328 39K views 3 years ago An explanation of the molecular geometry for the XeF4 (Xenon tetrafluroide) including a description of the XeF4 bond angles. The electron geometry for the Xenon.
Xef4 Vsepr
Chemistry tutorial for the Lewis dot structure and molecular geometry of xenon tetrafluoride (XeF4).

Leave a Comment Cancel Reply
1 Answer Stefan V. Aug 4, 2016 See explanation. Explanation: The first thing to do here is calculate how many valence electrons you have in one molecule of xenon tetrafluoride, XeF4. To do that, add the number of valence electrons that each atom brings to the table. You will have Xe: 8 e− aF: 7 e−
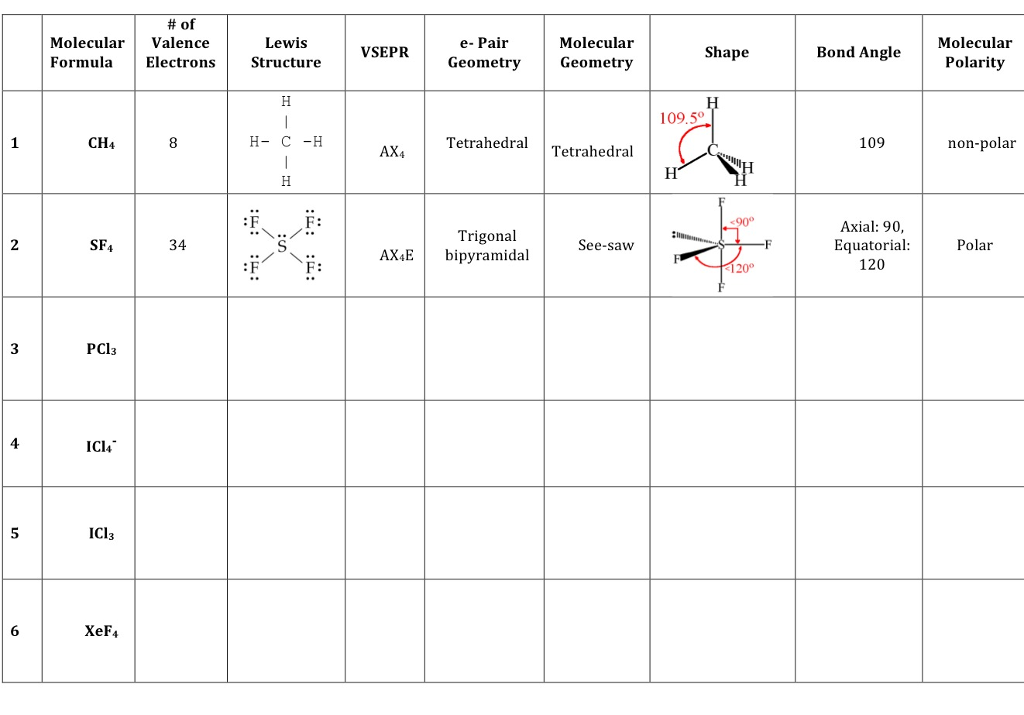
Solved Of Molecular Valence Formula Electrons CH4 SF4 34
The Lewis structure for XeF 4 requires you to place more than 8 valence electrons on Xe.. Let's do the XeF4 Lewis structure. Xenon has 8 valence electrons. Fluorine has 7, but we have four of the Fluorines; so that gives us 8 plus 28: 36 valence electrons. We'll put Xenon in the center, it's the least electronegative; and then Fluorines on.

XeF4 Lewis Structure, Molecular Geometry, Hybridization, and MO Diagram
XeF4 lewis structure involves one atom of xenon and four fluorine atoms. Xenon (Atomic number = 54 and electronic configuration = 2,8,18,18,8) belongs to group 18 of the periodic table and has 8 valence electrons.